Blood Journal - American Society of Hematology
Blood 2014 124:86
Sickle Cell Disease
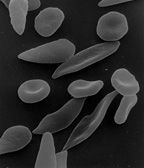
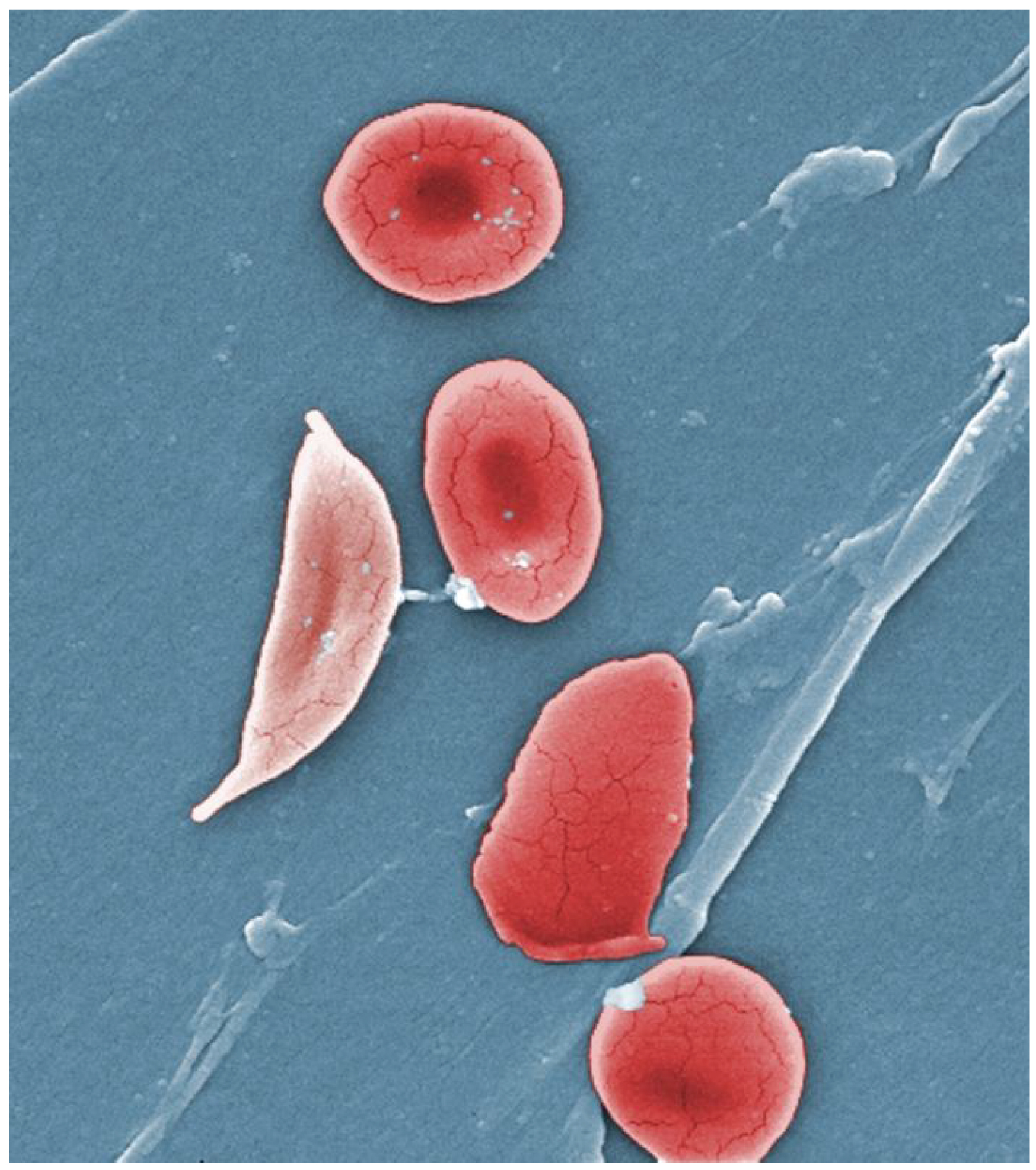
By OpenStax College - Anatomy & Physiology, Connexions Web site. http://cnx.org/content/col11496/1.6/, Jun 19, 2013., CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=30148180
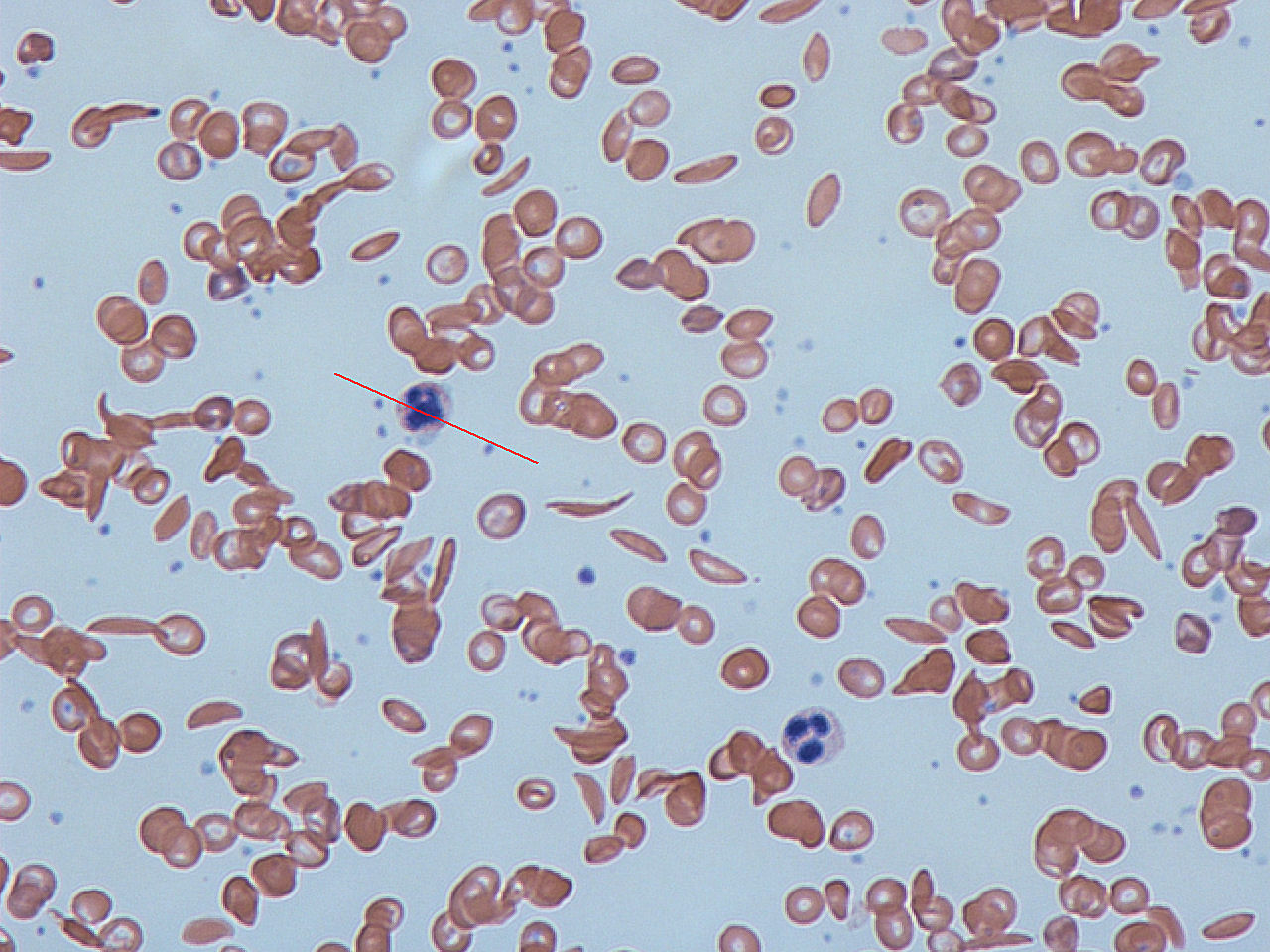
By Dr Graham Beards - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=18421017

single-nucleotide polymorphism (A to T) of the β-globin gene
glutamic acid (E/Glu) being substituted by valine (V/Val) at position 6. (7)
= HbS
under normal oxygen concentration = no effect
under conditions of low oxygen concentration = polymerization of the HbS
The deoxy form of haemoglobin exposes a hydrophobic patch on the protein between the E and F helices. The hydrophobic side chain of the valine residue at position 6 of the beta chain in haemoglobin is able to associate with the hydrophobic patch, causing HbS molecules to aggregate and form fibrous precipitates.
Vaso-Occlusive Crisis
The vaso-occlusive crisis is caused by sickle-shaped red blood cells that obstruct capillaries and restrict blood flow to an organ resulting in ischaemia, pain, necrosis, and often organ damage.
Acute Chest Syndrome
Acute chest syndrome is often precipitated by a lung infection, and the resulting inflammation and loss of oxygen saturation leads to further sickling of red cells, thus exacerbating pulmonary and systemic hypoxemia, sickling, and vaso-occlusion.
Splenic Sequestration Crisis
Splenic sequestration crises are acute, painful enlargements of the spleen, caused by intrasplenic trapping of red cells and resulting in a precipitous fall in haemoglobin levels with the potential for hypovolemic shock.
Aplastic Crisis
This crisis is normally triggered by parvovirus B19, which directly affects production of red blood cells by invading the red cell precursors and multiplying in and destroying them. Parvovirus infection almost completely prevents red blood cell production for two to three days. The shortened red cell life of SCD patients results in an abrupt, life-threatening situation. There is reticulocytopenia, and the rapid turnover of red cells leads to the drop in haemoglobin.
Background
Study
Method
Eligibility criteria included patients ≥ 5 years of age with documented diagnoses of HbSS or HbS/β0-thalassemia with at least two documented episodes of sickle cell crises (SCC) diagnosed in a medical facility during the 12 months prior to screening.
Pregnant women and patients with uncontrolled liver disease or renal insufficiency were excluded.
Prescription grade L-glutamine at 0.6 g/kg/day (max 30 g), or placebo, was taken in two divided doses. Daily dose was rounded to the closest 10 g.
The study drug was given for 48 weeks, then was tapered to zero over 3 weeks and a final evaluation visit was made 2 weeks after last dose.
The primary endpoint was number of SCC
Secondary endpoints include rates of hospitalization and adverse events; additional analyses include cumulative hospital days, incidence of acute chest syndrome (ACS) and time to first crises.
Results
Conclusion
Implication
| Weight (kg) | Dose per day (divided into two) |
|---|---|
| less than 30 | 10g |
| 30 to 65 | 20g |
| greater than 65 | 30g |
India?
The End (of Pain?)
Questions can be addressed to
Akshay S Dinesh
@asdofindia on twitter
Canonical source of this presentation: https://learnlearn.in/presentations/glutamine-sickle/